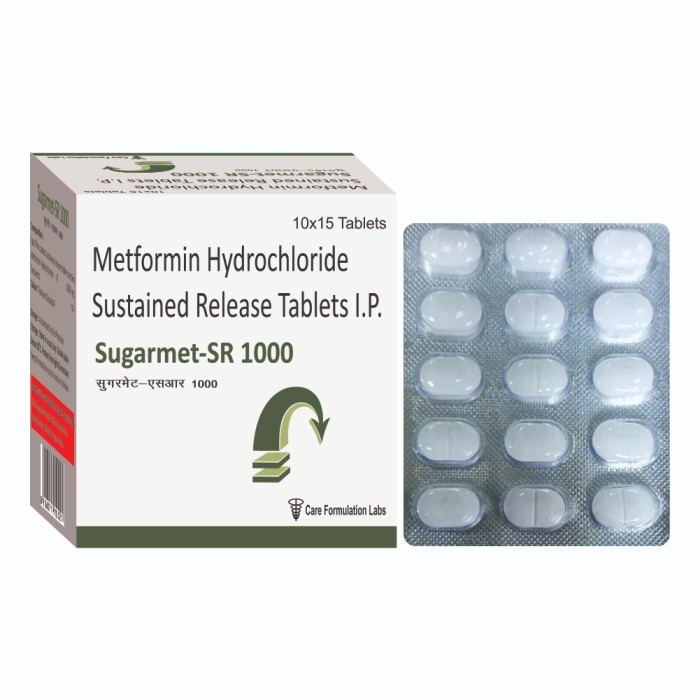
Metformin Hydrochloride IP 1000 mg tab
45.24 INR/Pack
Product Details:
- Solubility water
- Molecular Formula CHNOS
- Appearance White to off-white oval tablets with a light green sugar coating.
- Ingredients Metformin Hydrochloride IP 1000 mg, Sugar coating
- Feature Extended release formula, Enteric coated
- Shelf Life 2 Years
- Storage Instructions Store in Cool
- Click to View more
X
Metformin Hydrochloride IP 1000 mg tab Price And Quantity
- 45.24 INR/Pack
- 100 Pack
Metformin Hydrochloride IP 1000 mg tab Product Specifications
- White to off-white oval tablets with a light green sugar coating.
- CHNOS
- 2 Years
- Store in Cool
- Metformin Hydrochloride IP 1000 mg, Sugar coating
- Extended release formula, Enteric coated
- water
Metformin Hydrochloride IP 1000 mg tab Trade Information
- 5000 Pack Per Month
- 7 Days
Product Description
Description
- Each uncoated sustained release tablet contains: Metformin Hydrochloride IP 1000 mg + Excipients q.s.
Doses
- As Directed by Physician
Indication
- Metformin is an oral diabetes medicine that helps control blood sugar levels. Metformin is used together with diet and exercise to improve blood sugar control in adults with type 2 diabetes mellitus. Metformin is sometimes used together with insulin or other medications, but it is not for treating type 1 diabetes
Advanced Extended Release Formula
These Metformin Hydrochloride IP 1000 mg tablets utilize an extended release mechanism and enteric coating, providing a controlled and consistent release of medication. This approach helps maintain stable blood sugar levels over longer periods, reducing the frequency of dosing and minimizing side effects associated with immediate-release formulations.
Easy Storage and Shelf Life
To ensure maximum efficacy, store the tablets in a cool environment away from excessive moisture and direct sunlight. The product retains its potency for up to two years, offering reliability for both individual users and distributors managing large inventories.
Widely Available and Trusted Manufacturing
Produced and distributed across India by reliable manufacturers and suppliers, including importers, exporters, wholesalers, and retailers, these tablets are accessible to healthcare providers and patients nationwide. Their quality and consistent availability make them a preferred choice for blood glucose management.
FAQ's of Metformin Hydrochloride IP 1000 mg tab:
Q: How should Metformin Hydrochloride IP 1000 mg tablets be taken for optimal results?
A: It is recommended to take these extended release tablets with food, usually once daily or as prescribed by your healthcare provider, to maximize absorption and reduce gastrointestinal side effects.Q: What are the primary benefits of the extended release and enteric-coated formula?
A: The extended release and enteric coating ensure steady medication levels, improve gastrointestinal tolerance, and allow for fewer daily doses, enhancing patient compliance and comfort.Q: When do these Metformin tablets start to take effect?
A: While individual responses may vary, blood glucose-lowering effects typically begin within a few days of starting therapy, with full benefits seen after consistent use as directed by a physician.Q: Where are these tablets manufactured and distributed?
A: These Metformin Hydrochloride IP 1000 mg tablets are produced in India and are distributed through a wide network of dealers, exporters, wholesalers, and retailers across the country.Q: What is the process for storing Metformin Hydrochloride IP 1000 mg tablets to maintain their quality?
A: Store the tablets in a cool, dry place, away from excessive heat and moisture. Keeping them in their original packaging will help preserve their shelf life of up to two years.Q: How does the molecular formula C10H15NO5S relate to the product's function?
A: The molecular formula C10H15NO5S represents the chemical composition of Metformin Hydrochloride, which is responsible for the tablet's glucose-lowering action and solubility characteristics.Tell us about your requirement

Price:
Quantity
Select Unit
- 50
- 100
- 200
- 250
- 500
- 1000+
Additional detail
Mobile number
Email
Other Products in 'Anti Diabetic Medicine' category
We accept minimum 10 boxes of all products.


 Send Inquiry
Send Inquiry
 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese